12 MIN READ
Ear Rots, Molds, and Mycotoxin Management
October 19, 2023
- Mycotoxins produced by ear rots and molds affect grain quality and end use.
- Identifying the fungi infecting kernels can help growers determine if additional precautions need to be taken to stop mycotoxin production.
- Corn grain must be stored at proper moistures and temperatures to preserve its storage life.
Diagnosing Ear Rots and Molds
Diagnosing ear rots and molds is important because they can produce mycotoxins that are harmful and potentially deadly to humans and livestock, and because diseased kernels can reduce test weight, grain quality, and yield potential. Some ear rots require hot, dry weather to develop, while others are associated with wetter and cooler conditions. Wounds created by insects, mechanical damage, and hail can provide openings for pathogen to infect the kernel.
If ear rots are found during routine scouting, infected fields should become a harvest priority. Infected grain should be tested by a toxicology lab to determine if mycotoxins are present. If mycotoxins are present, appropriate marketing and/or feeding measures should be instituted based on FDA guidelines. Please see What you Need to Know about Aflatioxin for additional information on mycotoxins associated with ear rots.
Moisture and Temperatures for Storage
A single day of holding onto warm, wet corn before proper drying can result in a 50 percent loss of storage life.1 Expect to harvest infected grain at higher moisture content and then dry it to below 14% total moisture content within 48 hours. Lower temperatures also slow mold growth and mycotoxin development. Storage life can be lengthened by quickly drying grain and cooling it to 40 °F or colder.1
Corn Ear Rots and Molds
Aspergillus ear rot (Aspergillus species): A gray-green or light-green powdery mold starting at the ear tip (Figure 1). Development is favored by hot, dry conditions after damage to silks or kernels from insects, hail, birds, or other factors. Aspergillus can be a grain-storage mold and an issue in the field because of its ability to grow at 15% moisture content.2,3 These fungi are capable of producing aflatoxin, a mycotoxin toxic to livestock and humans. Aflatoxin production increases with nitrogen stress and temperatures above 80 °F.4 Fields with severe Aspergillus ear rot should be prioritized for earlier harvest with the grain being dried to below 15% moisture for harvest.

Cladosporium kernel rot (Cladosporium herbarum): A gray to black or very-dark-green powdery mold that may appear in streaks scattered across the ear (Figure 2). The mold can be rubbed off the kernel surface. Infects kernels are often those damaged by insects, hail, frost, and other factors. The disease is favored by wet weather during grain fill. However, it is more common in hot, dry years. C. herbarum can grow at temperatures greater than 90 °F and grain moisture content as low as 16%.5
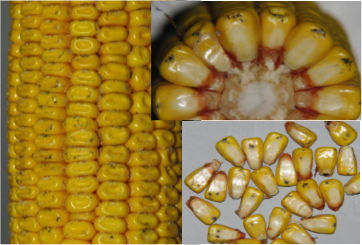
Diplodia ear rot (Stenocarpella maydis): A white to gray mold that can cause the entire ear to appear brown (Figure 3). Infection can begin at the ear base and move toward the tip, growing between kernels, but infection can also start at the tip of the ear, especially following insect or bird damage. Pycnidia, or fruiting bodies, can form on husks and at the base of kernels and can help in distinguishing Diplodia from other ear rots. Under severe infection, the ear may be entirely covered by white mycelium that, “mummifies” the ear and may cause the husk to stick to the kernels. Dead husks and ear leaves may be one of the first indications of Diplopia in the field. Cobs can also be infected via the ear shank, causing yield loss due to poorly developed ears and/or kernels. Diplodia ear rot occurs most often in reduced tillage and continuous corn systems. Development of this disease is favored by warm, dry conditions prior to silking followed by wet conditions after silking. S. maydis is not known to produce mycotoxins, but reports from other countries have associated this fungus with diplodiosis—a condition involving a lack of voluntary muscle coordination, muscle weakness, or partial paralysis—in cattle and sheep.6

Fusarium ear rot (Fusarium species): A white to pink mold which infects random kernels around the ear, and which may cause a starburst pattern on kernel caps (Figure 4). Fusarium infection may occur from late vegetative growth stages to three weeks after midsilk.3 Fusarium can infect a kernel through growth cracks or damage, such as that from insects, hail, or other mechanical injuries. Warm, wet weather after silking favors development of Fusarium ear rot. Fusarium can produce the mycotoxins zearalenone; fumonisin, which is toxic to livestock, particularly horses; and vomitoxin, also known as deoxynivalenol (DON).6,7

Gibberella ear rot (Gibberella zeae): The coloration is often bright pink, but it varies from red to white. Gibberella ear rot usually begins at the ear tip and progresses toward the base (Figure 5). Ears become “mummified” because the pink or white mycelial growth completely covers the ear and causes kernels to stick to husks and cobs. Favorable conditions for the development of Gibberella ear rot include cool temperatures and wet weather within three weeks after silking.4 Potential mycotoxins include vomitoxin, also known as deoxynivalenol (DON), and zearalenone, which may cause sickness or death in livestock.7

Nigrospora ear rot or cob rot (Nigrospora sphaerica, synonym N. oryzae): Infected kernel tips and cob pith can be covered with very small, jet-black spore masses (Figure 6). Kernels appear bleached, often have whitish streaks starting at the tips and extending toward the crowns, and may have a gray mold growth. Infected ears are easily broken into small pieces during harvest because of the rotted pith (Figure 7). Infection usually starts at the ear butt. This infection is often associated with premature plant death from frost, hail, or drought, and from leaf, stalk, and root diseases. The disease may appear more often in low fertility situations.4


Penicillium ear rot or “Blue Eye” (Penicillium species): A powdery blue-green mold which grows between kernels, usually at the tip of the ear (Figure 8). Infection generally occurs on kernels damaged by hail, insects, frost, or other factors, though Penicillium is capable of infecting non-injured kernels with 14% moisture content.3 Penicillium is an important grain storage mold. Affected grain should be dried to 13 to 15% moisture content, depending on storage duration, to prevent further damage.8 These fungi can produce mycotoxins.7

Trichoderma ear rot (Trichoderma viride): A dark-greenish mold which grows on and between husks and kernels. Sprouting can occur when the infection is severe (Figure 9). Trichoderma ear rot development is favored by excessive rainfall and damage from insects, hail, or other mechanical injuries to the ear. The disease occurs more often occurs when there is above-average rainfall the month before harvest.3

Management of Crop Stress on Stalk Health
Best management practices (BMPs) for ear rot management should begin prior to planting, because little can be done to stop a progressing infection once it has established itself on ears in fields. Strategies that help minimize crop stress and disease starting in stalks can help reduce the potential for ear rot development.
Factors that can help reduce crop stress include:
- Test soil and fertilize accordingly.
- If possible, irrigate to help reduce drought conditions.
- Use insect-protected corn products (particularly for European corn borer, earworm, and western bean cutworm).
- Use foliar disease-resistant corn products.
- Use fungicides to help manage foliar diseases.
- Avoid continuous corn.
- Utilize tillage to manage infected residue.
Scout fields before harvest and be ready to harvest earlier at a higher moisture to stop further kernel infection and potential mycotoxin production. Grain coming off fields with mold can be expected to have a storage only life 60 to 70% that of ‘clean’ grain.1 Seed conditioning and grain cleaning can be considered if loads have high mycotoxin levels.
Sources:
1 Hurburgh, C. 2016. Wet weather creates challenges for harvest. Iowa State University Extension and Outreach. Integrated Crop Management News. Wet Weather Creates Challenges for Harvest | Integrated Crop Management (iastate.edu).
2 Woloshuck, C. and Wise, K. 2011. Diseases of corn: Aspergillus ear rot. Purdue Extension. BP-83-W. https://www.extension.purdue.edu/extmedia/bp/bp-83-w.pdf
3 Thomison, P., Lohnes, D., Geyer, A., and Thomison, M. Troubleshooting abnormal corn ears. The Ohio State University. https://u.osu.edu/mastercorn/
4 1991. Corn ear and kernel rots. University of Illinois. Integrated Pest Management Reports on Plant Diseases. RPD No. 205. http://ipm.illinois.edu.
5 2019. Cladosporium ear rot of corn. Crop Protection Network. https://cropprotectionnetwork.org/encyclopedia/cladosporium-ear-rot-of-corn
6 Muillo-Williams, A., Collins, A., Esker, P.D. 2022. Corn ear rots and mycotoxins. PennState Extension. https://extension.psu.edu/corn-ear-rots-and-mycotoxins.
7 Jackson-Ziems, T.A., Giesler, L.J., Harveson, R.M, Korus, K.A., Liu, B., Wegulo, S.N. 2012. Corn Disease Profile III corn ear rot diseases and grain molds. University of Nebraska-Lincoln. EC1901. https://extensionpublications.unl.edu/assets/pdf/ec1901.pdf
8 Munkvold, G.P. and White, D.G. (Eds.) 2016. Compendium of corn diseases, fourth edition. The American Phytopathological Society. https://apsjournals.apsnet.org/doi/book/10.1094/9780890544945
1211_150874
Disclaimer
Always read and follow pesticide label directions, insect resistance management requirements (where applicable), and grain marketing and all other stewardship practices.