17 MIN READ
Identification of Cotton Diseases
March 23, 2021
Get Year-Round Updates From Our Agronomic Experts
Reducing the impact of disease throughout the season begins with crop management planning. Understanding the disease history of the field can help with variety selection and crop rotation. Including a seed treatment can help provide broad-spectrum protection for cotton seedings. Fields that are under conservation tillage can be slower to warm up and more prone to overwintering pathogens. Plant when soil temperatures are above 65°F at a 4-inch depth, with a favorable 5-day weather forecast and do not plant seeds too deep. A depth of 0.5 inches is ideal under good growing conditions. Scouting for disease should begin early and coincide with insect scouting. Solid management decisions are based on knowing what and how many pests are present.
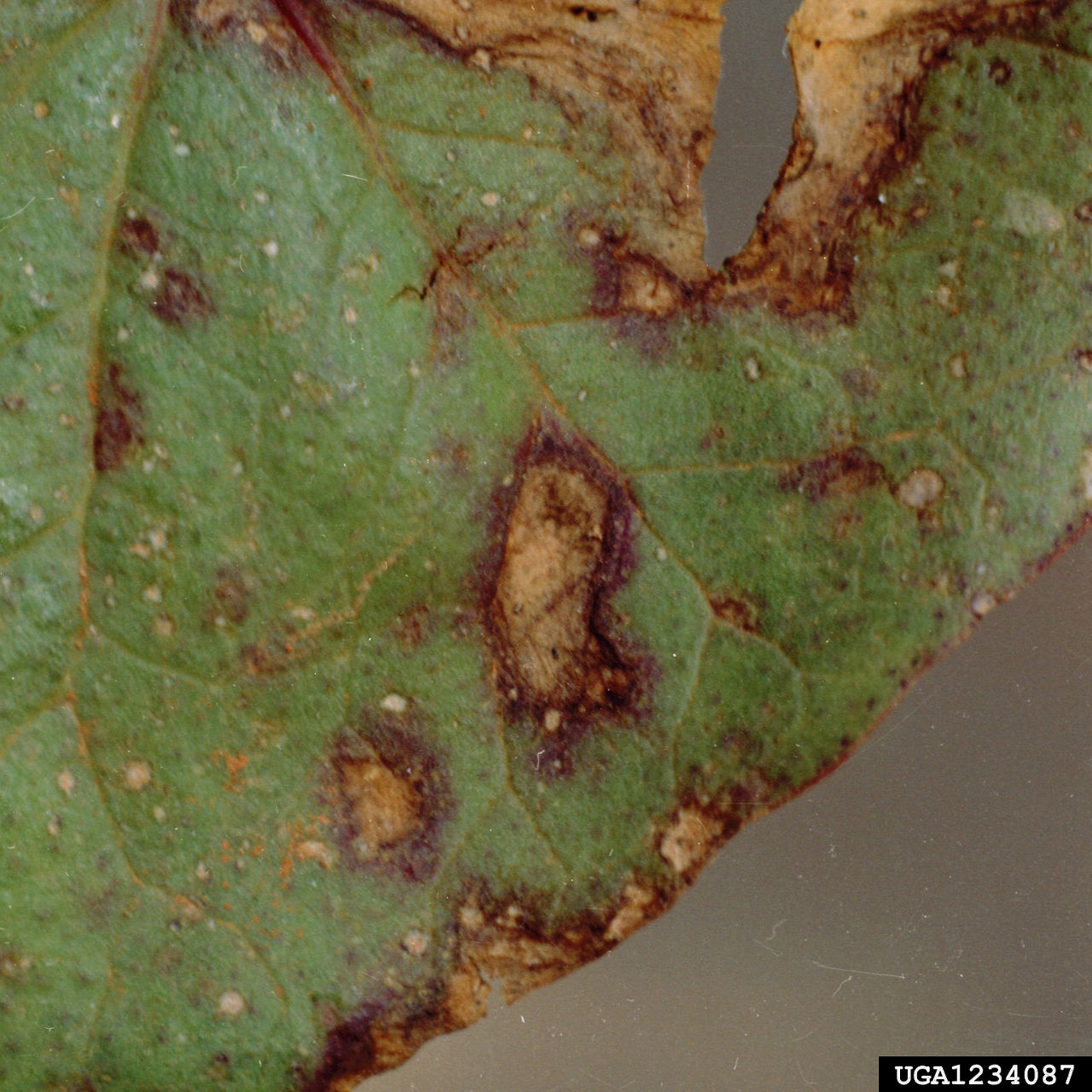
Mid- to late-season cotton diseases occur when a crop is under stress. Stress can come from all directions—too dry, too wet, insects, storms, fertility, rank growth, and others all play a part in the health of a crop. Mitigating conditions conducive for disease development and understanding if and when a fungicide application is necessary is important to help maximize yield potential.
Wilt diseases can be difficult to diagnose and manage, but proper fertility management, crop rotation to corn or soybean, use of nematicides, and cotton variety selection can help reduce yield losses from wilt diseases.
Nematode damage has historically been attributed to poor fertility or poor soil conditions. Over time, a better understanding has led to identification of nematodes and the yield losses they can cause. Nematodes reduce cotton vigor by injuring roots, which limits root growth, leaving cotton plants susceptible to environmental stress, insect damage, and diseases. Cultural and chemical control can help reduce the impact of nematodes on cotton yield potential.
Click the tabs below to indentify cotton diseases that may impact cotton crops.
Seedling Diseases
Foliar Diseases
Stem and Root Diseases
Rust
Boll Rots
Nematodes
Diseases of Unknown Cause
Sources:
1 Thiessen, L. 2019. Disease management in cotton. 2019 Cotton Information. North Carolina State University. https://content.ces.ncsu.edu/cotton-information/disease-management-in-cotton.
2Whitaker, J., Culpepper, S., Freeman, M., Harris, G., Kemerait, B., Perry, C., Porter, W., Roberts, P., Liu, Y., and Smith, A. 2018. 2019 UGA Cotton Production Guide. The University of Georgia.
3Texas Plant Disease Handbook. Cotton Gossypium hirsutum. Texas A&M AgriLife Extension. https://plantdiseasehandbook.tamu.edu/industry-specialty/fiber-oil-specialty/cotton/.
4Cotton seedling disease identification. The National Cotton Council. https://www.cotton.org/tech/pest/seedling/identification.cfm.
5Isakeit, T. 2016. Managing Seedling Diseases of Cotton. Texas A&M University. http://agrilifecdn.tamu.edu/coastalbend/files/2017/06/Management-of-seedling-diseases-of-cotton_2016.pdf.
6Cotton foliar diseases. The University of Tennessee Institute of Agriculture. https://guide.utcrops.com/cotton/cotton-foliar-diseases/.
7Woodward, J. 2016. Increased incidence of Alternaria leaf spot of cotton. Texas A&M AgriLife Extension. https://agrilife.org/texasrowcrops/2016/09/06/increased-incidence-of-alternaria-leaf-spot-of-cotton/.
8McGriff, E. 2016. Stemphylium leaf spot in cotton. Alabama Cooperative Extension System. https://sites.aces.edu/group/timelyinfo/Lists/Posts/Post.aspx?ID=836.
9Thiessen, L. 2018. Areolate mildew confirmed in North Carolina. North Carolina State Extension. https://plantpathology.ces.ncsu.edu/2018/08/areolate-mildew-confirmed-in-north-carolina/.
10Nichols, K. 2019. Target spot, areolate mildew damaging to cotton. Alabama A&M and Auburn Universities Extension. https://www.aces.edu/blog/topics/cotton/target-spot-areolate-mildew-damaging-to-cotton/.
11Tabassum, A., Bag, S., Roberts, P., Suassuna, N., Chee, P., Whitaker, J.R., Conner, K.N., Brown, J., Nichols, R.L., and Kemerait, R.C. 2019. First report of cotton leafroll dwarf virus infecting cotton in Georgia, U.S.A. The American Phytopathological Society. https://apsjournals.apsnet.org/doi/10.1094/PDIS-12-18-2197-PDN.
12Schattenberg, P. 2019. Cotton leafroll dwarf virus discovered in Central Texas. Texas A&M AgriLife. https://today.agrilife.org/2019/08/23/cotton-leafroll-dwarf-virus-discovered-in-central-texas/.
13Isakeit, T. and Morgan, G. 2018. Limiting the spread of fusarium wilt race 4, a new disease of cotton in Texas. Texas AgriLife Extension. https://agrilife.org/texasrowcrops/2018/09/11/limiting-the-spread-of-fusarium-wilt-race-4-a-new-disease-of-cotton-in-texas/.
14Raper, T., Meyer, B., Lawrence, K., Sandlin, T., Cutts, T., Silvey, N., Burmester, C., Dill, T., Shelby, P., and Kelly, H. 2017. Verticillium wilt in Tennessee valley cotton. W403. http://news.utcrops.com/wp-content/uploads/2017/03/W403.pdf.
15Isakeit, T. 2016. Cotton root rot (Phymatotrichopsis root rot) and its management. Texas AgriLife Extension. PLPA-FC010-2016. https://www.cottoninc.com/wp-content/uploads/2018/01/Cotton-root-rot-phymatotrichopsis-management.pdf.
16Goldberg, N. and French, J. Southwestern cotton rust. New Mexico State Extension Plant Pathology. https://aces.nmsu.edu/ces/plantclinic/documents/southwester-cotton-rust-nycu.pdf.
17Cutts, T. 2016. Cotton boll rot: potential causes and management considerations. Alabama Cooperative Extension System. https://sites.aces.edu/group/crops/blog/Lists/Posts/Post.aspx?List=93b27053-563c-4a66-b5db-09c41227b355&ID=139&Web=89eff7f6-d4b3-4cf8-9191-103ef5cc38d3.
18Thiessen, L. and Rivera, Y.R. 2019. Root knot nematode of cotton. NC State Extension. https://content.ces.ncsu.edu/cotton-root-knot-nematodes.
19Overstreet, C. 2016. Nematode problems on cotton. LSU College of Agriculture. https://www.lsuagcenter.com/profiles/coverstreet/articles/page1460046020798.
20Blasingame, D., Gazaway, W., Kemerait, R., Kirkpatrick, T., Koenning, S., Lawrence, G., McClure, M., Mueller, J., Newman, M., Overstreet, C., Phipps, P., Rich, J., Thomas, S., Wheeler, T., and Wrather, A. 2003. Cotton nematodes - your hidden enemies. Beltwide Cotton Nematode Research and Education Committee. http://cotton.tamu.edu/Nematodes/nematodebrochure.pdf.
21Crow, W.T. 2015. Sting nematode. University of Florida. http://entnemdept.ufl.edu/creatures/nematode/sting_nematode.htm.
22Bell, A.A., Nichols, R.L., Albers, D., Baird, R., Brown, S., Colyer, P., El-Zik, K., Gwathmey, C.O., Lemon, R., Newman, M., Phipps, B.J., and Oosterhuis, D.M. 2002. Texas Cooperative Extension. L-5412. http://cotton.tamu.edu/Nematodes/bronzewilt.pdf.
Web sources verified 10/11/2019, 1009_G1
Disclaimer
Always read and follow pesticide label directions, insect resistance management requirements (where applicable), and grain marketing and all other stewardship practices.